Transcatheter Intervention for Infantile Hepatic Hemangioendothelioma with Amplatzer Duct Occluder II
———————I. B. Vijayalakshmi1*, Narasimhan Chitra1, Mallikarjun Kavya1, Kumar Basant1, Hegde Madhav1 and C. N. Manjunath1
ABSTRACT
Introduction: Infantile Hepatic hemangioendothelioma is characterized by multifocal
benign vascular dilatations involving the liver. The clinical course depends on tumor size,
localization and complications.
Case Report: 15 days old neonate with stormy postnatal period was referred for
intractable congestive cardiac failure. Transthoracic 2D echocardiography showed
multiple vegetations on all the four valves, noncompaction of left ventricle with multiple
vascular channels in left lobe of liver. Computed tomography angiogram showed
58x29x50 mm markedly enhancing lesion in left lobe of liver suggestive of infantile
hepatic hemangioendothelioma. The neonate was treated for bacterial endocarditis. Later
6x6 Amplatzer duct occluder II was parked in hepatic vein, then gel foam and polyvinyl
chloride particles were injected into infantile hepatic hemangioendothelioma. Cardiac
failure resolved with marked regression of lesion.
Discussion: Infantile hepatic hemangioendothelioma is a rare anomaly causing heart
failure in neonate causing death in up to 70% of untreated infants without adequate
regression of lesion. Therefore aggressive treatment is warranted. For the first time in the
world, we report a case of infantile hepatic hemangioendothelioma in a neonate, with
pump failure due to noncompaction of left ventricle and vegetations on all four valves,
successfully treated by combination of transcatheter deployment of Amplatzer duct
occluder II to occlude the venous end and hand injection of gel foam and polyvinly
chloride particles from the aortic end to close the feeder artery.
Conclusion: The combination of closure of hepatic vein by Amplatzer duct occluder II
and injection of gel foam and polyvinyl chloride particles is safe and effective in infantile
hepatic hemangioendothelioma in neonate with heart failure.
Keywords: Neonatal endocarditis; vegetations; ADO II; gel foam; PVC particles; LV
noncompaction; intractable heart failure
1. INTRODUCTION
Infantile Hepatic hemangioendothelioma (IHH) is a rare congenital syndrome characterized by multifocal benign vascular dilatations involving the liver. The clinical course depends on tumor size, its growth characteristics, the localization, and complications. The tumor may be treated conservatively with interferon, corticosteroid (controversial and contraindicated in our case), cytotoxic agents, and irradiation which are hazardous. The radical treatment such as hepatic artery ligation, hepatic artery embolisation with steel coils, polyvinly alcohol injection and surgical resection or even liver transplantations are reported in literature. These conventional managements carry very high risk; hence we adopted non surgical highly successful, safe and effective transcatheter management of IHH. To the best of our knowledge this is the first case in the world in which the combination of transcatheter deployment of Amplatzer duct occluder II (ADO II) with grated gel foam and polyvinly chloride (PVC) particles were used to achieve successful closure of the shunt.
2. CASE REPORT
A 15 days old male neonate was referred with history of chest retractions, not maintaining oxygen saturation and feeding difficulties. This child was delivered by caesarian section at 36 weeks of gestation for premature rupture of membranes of two days duration. The birth weight was 3.15 kgs. The child developed sepsis, heart failure, acute tubular necrosis, hypoglycemia and hyperbilirubinemia. The transthoracic echocardiography (TTE) done at 15 days of age showed noncompaction of left ventricle with reduced function (EF-40%), pulmonary hypertension (50 mm Hg), multiple vegetations on all four valves (Figs. 1A,B,C,D) and multiple vascular channels in the left lobe of liver with both arterial and venous pattern, which was suggestive of vascular malformation. The CT angiogram of the abdomen showed 58x29x50 mm markedly enhancing lesion seen in relation to left lobe of liver suggestive of haemangioendothelioma. As per the blood culture report (Gram negative bacilli-Klebsiella) the neonate was treated with the antibiotics meropenam 100 mgs twice a day for three weeks and ceftriaxone 150 mg twice a day for four weeks.
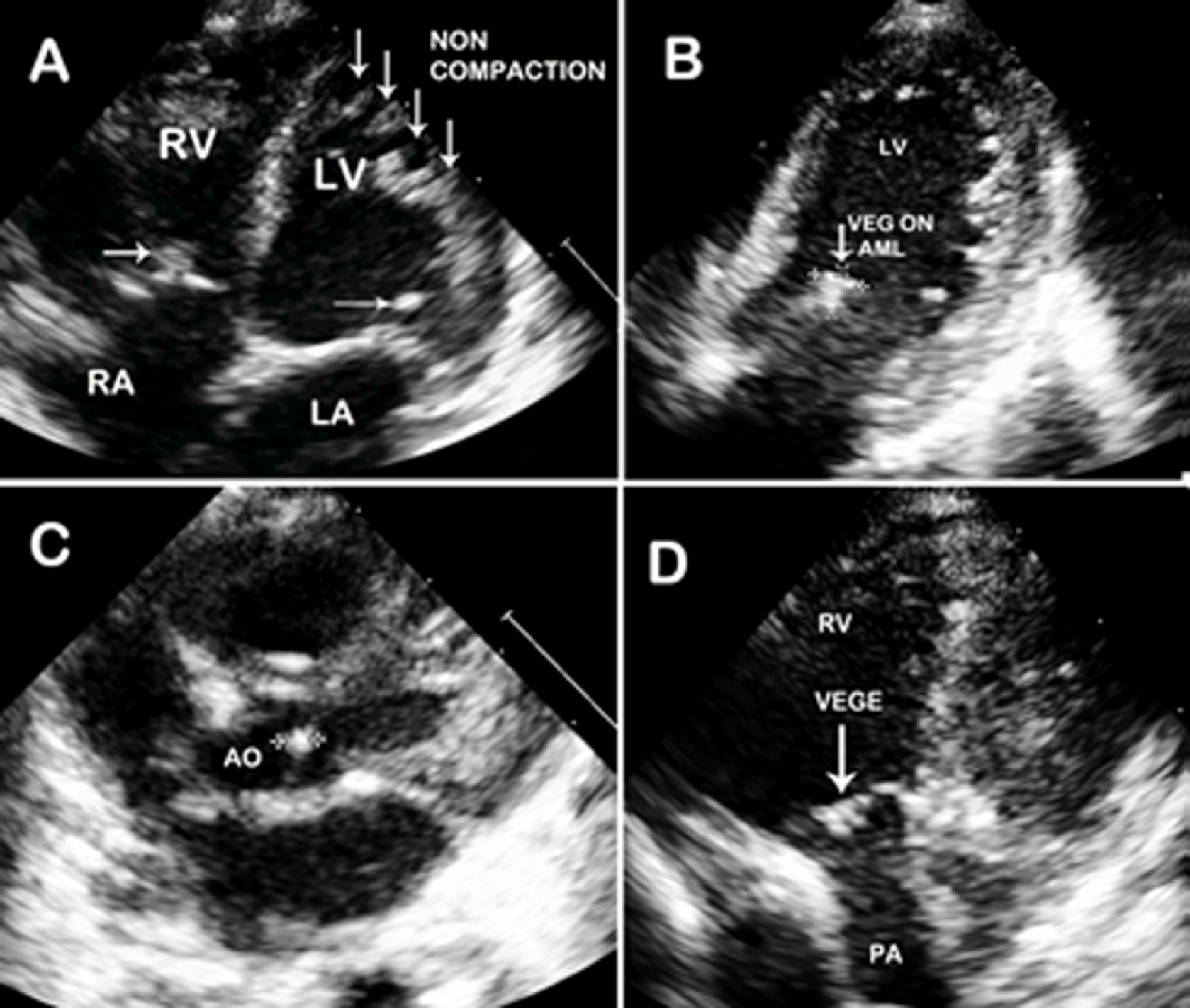 Fig. 1A. Transthoracic 2D echocardiography in apical four chamber view shows
vegetation on tricuspid valve and mitral valve with deep recesses due to LV non
compaction. B: Modified two chamber view shows large vegetation on the anterior
mitral leaflet. C: Short axis shows vegetation on aortic valve. D: Modified view shows
multiple fuzzy echoes on pulmonary valve
Fig. 1A. Transthoracic 2D echocardiography in apical four chamber view shows
vegetation on tricuspid valve and mitral valve with deep recesses due to LV non
compaction. B: Modified two chamber view shows large vegetation on the anterior
mitral leaflet. C: Short axis shows vegetation on aortic valve. D: Modified view shows
multiple fuzzy echoes on pulmonary valve
The procedure was done on 40th day of life. Both left and right heart catheterization was done through right femoral artery and vein by Seldinger’s technique under epidural anesthesia and intravenous ketamine. The descending aorta angiogram done in anterioposterior view with 4F pigtail catheter showed highly vascular lesion, fed through the celiac plexus draining into grossly dilated (10 mm) hepatic vein (Fig. 2A). Then with 4F Judkins right coronary artery (RCA) catheter, selective angiogram was done to identify the feeders. There were two feeders. The bigger of the two arteries with circuitous course was supplying the major portion of the mass. The lesion was entered using 0.018” x 260 cms Terumo guide wire and 4F Terumo catheter was passed over the wire, well into the middle of the lesion. Through the venous sheath, 0.018” x260cms Terumo guide was passed into the dilated hepatic vein. Over this curved Cooks 4F shuttle sheath was inserted into the hepatic vein (Fig. 2B). Then through the shuttle sheath, 6 x 6 ADO II device (St Jude Medical, Minnesota) was deployed at the bend of the vessel, but not released (Fig. 2C ). The grated gel foam particles mixed with diluted contrast were injected from the arterial end through 4F
Terumo catheter. Almost complete cessation of flow through the anomalous channel was achieved by instillation of PVC particles at the end. The care was taken so that the diluted contrast containing PVC particles did not reflux into the celiac plexus. Once the check angiogram showed good occlusion of the mass, the ADO II device was released. Post procedure angiogram showed no flow into the hepatic lesion from the arterial end and the device was in good position at the venous end (Fig. 2D). The highly vascular lesion before procedure (Fig. 3A) regressed completely post procedure with the device in situ (Fig. 3B). The patient developed distension of upper abdomen after the procedure. This was treated conservatively as peristaltic sounds were well heard and the patient recovered within 48 hours. Post procedure there was also a local complication with loss of pulse in right femoral artery. This was treated with intravenous heparin infusion for 24 hours followed by intravenous heparin 8th hourly for 72 hour, till the pulse was well felt and there were good Doppler signals. This led to reappearance of vascular channels in the lesion on abdominal ultrasound. But the CT angiogram done after 3 months showed significant reduction in the size of the mass from 58x29x50 mm (before procedure) to 25 x 10 x 20 mm (Fig. 4A and B). The child has gained weight (5.25kgs), review echocardiogram showed hyper echogenic healed vegetations, not mobile and EF improved to 55%.
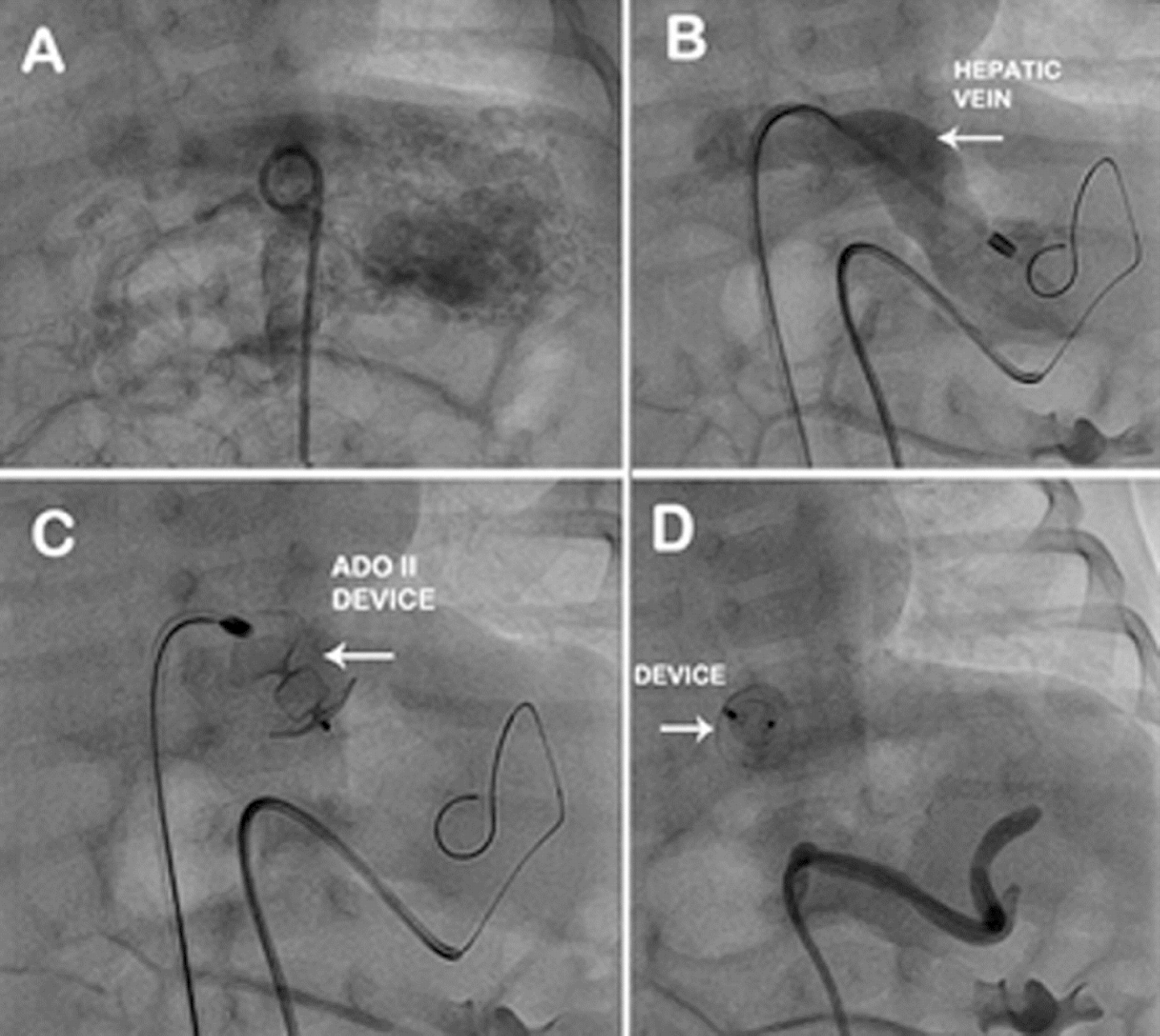 Fig. 2A. Abdominal aortogram with pig tail catheter shows IHH.B: 4 F Terumo catheter
with the Terumo guide wire in the mass and 4 F Cooks shuttle sheath through inferior
vena cava into dilated left hepatic vein. C: Amplatzer Duct Occluder II device with two
retention discs and middle lobe parked in the hepatic vein. D: Check angiogram
shows complete occlusion of IHH with Amplatzer Duct Occluder II in situ
Fig. 2A. Abdominal aortogram with pig tail catheter shows IHH.B: 4 F Terumo catheter
with the Terumo guide wire in the mass and 4 F Cooks shuttle sheath through inferior
vena cava into dilated left hepatic vein. C: Amplatzer Duct Occluder II device with two
retention discs and middle lobe parked in the hepatic vein. D: Check angiogram
shows complete occlusion of IHH with Amplatzer Duct Occluder II in situ
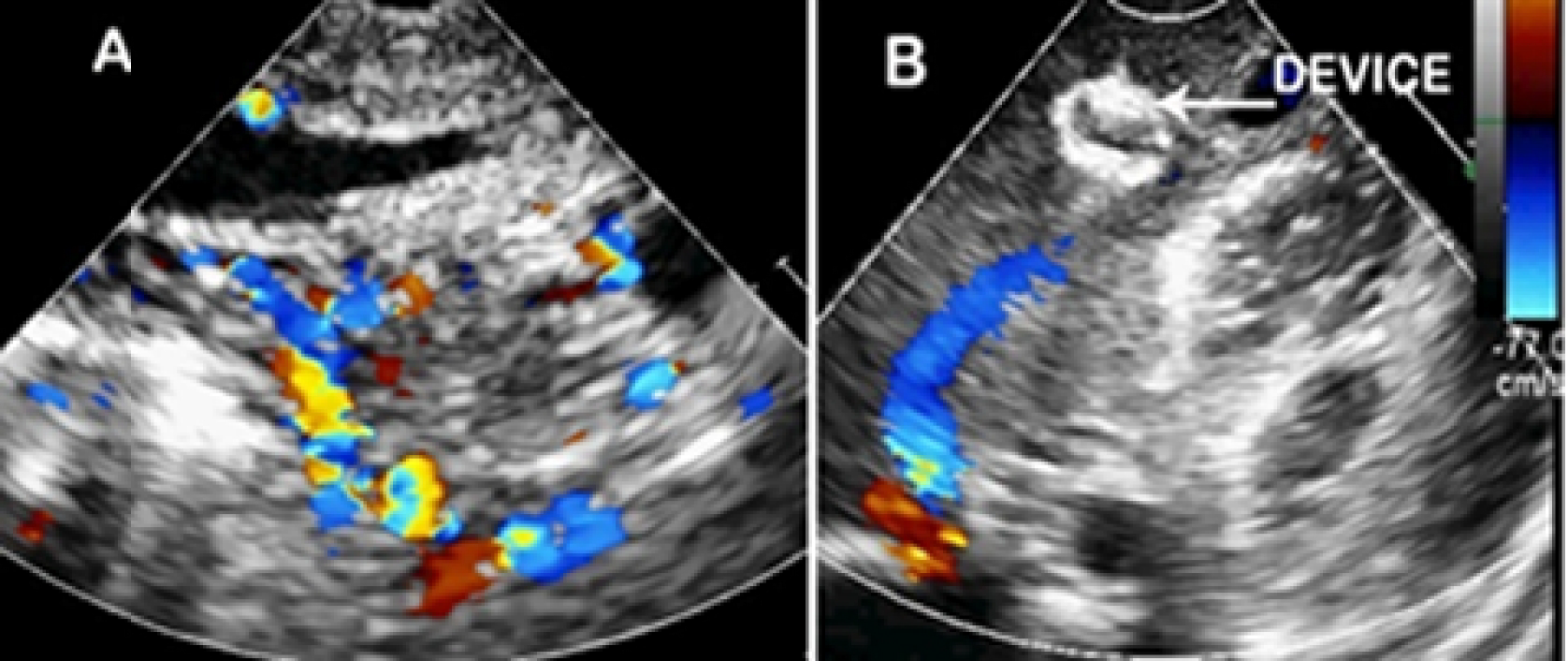 Fig. 3A. Transthoracic 2-D echocardiography with color Doppler shows multiple
vascular channels in the left lobe of the liver with both arterial and venous pattern. B:
Post intervention echocardiography with color Doppler shows device in situ with
disappearance of vascular channels and marked reduction in the size of hepatic vein
Fig. 3A. Transthoracic 2-D echocardiography with color Doppler shows multiple
vascular channels in the left lobe of the liver with both arterial and venous pattern. B:
Post intervention echocardiography with color Doppler shows device in situ with
disappearance of vascular channels and marked reduction in the size of hepatic vein
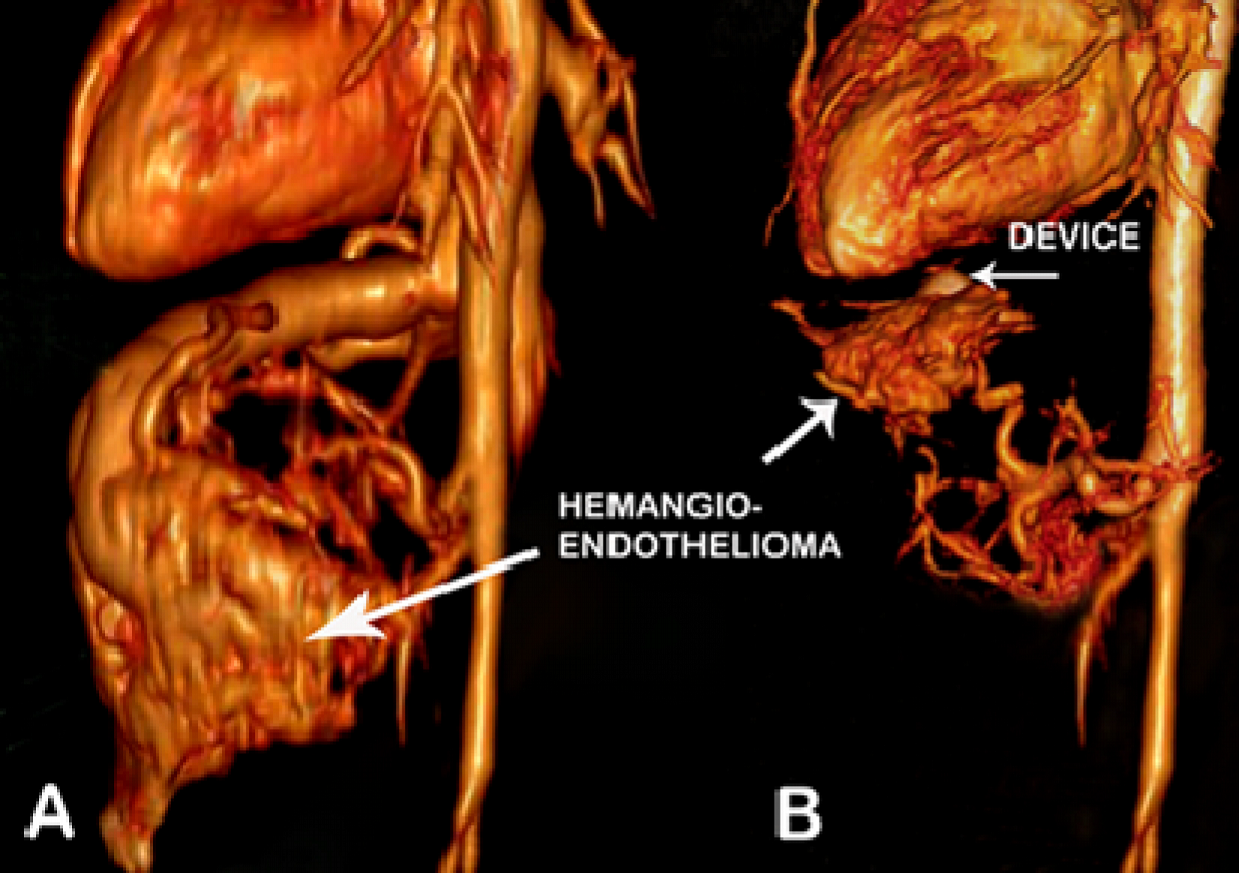 Fig. 4A. Volume rendered 3D reconstructed CT angiogram image shows large
hemangioendothelioma of left lobe of the liver draining into grossly dilated hepatic
vein B: Volume rendered 3D reconstructed CT angiogram image on follow up after 3
months shows device (arrow) in situ, mass has regressed and hepatic vein has
completely disappeared
Fig. 4A. Volume rendered 3D reconstructed CT angiogram image shows large
hemangioendothelioma of left lobe of the liver draining into grossly dilated hepatic
vein B: Volume rendered 3D reconstructed CT angiogram image on follow up after 3
months shows device (arrow) in situ, mass has regressed and hepatic vein has
completely disappeared
3. DISCUSSION
Infantile hepatichemangioendothelioma is a rare congenital anomaly. The incidence is 1/20,000. It is basically a capillary hemangioma which consists of a network of capillary sized and endothelium lined vessels. The clinical manifestations of IHH are variable. The most feared complications associated with this disease include high cardiac output congestive heart failure, consumptive coagulopathy and thrombocytopenia (Kasabach- Merritt syndrome) [1]. Rapid AV shunting through vascular channels leads to an increased cardiac output. The dilation of hepatic arteries and recirculation of blood back to right heart may lead to overt heart failure (58%) [2,3]. Heart failure can cause death in up to 70% of untreated infants without adequate regression of the lesion [4,5]. Therefore, aggressive treatment is warranted.
In the literature there are reports of tumor being treated conservatively with interferon [2], corticosteroid [4,6], cytotoxic agents [7], and irradiation [8] and alcohol injection. The radical treatment such as hepatic artery ligation [5,9-11], surgical resection [5,12,13], or even liver transplantation are reported. The precise mechanism of action of steroids is unclear. Side effects are hypertension, hyperglycemia, sepsis and pseudo tumor cerebri especially when it is given to infants [5]. In our case steroids were contraindicated as patient had bacterial endocarditis of all the four valves. In this 15 days old neonate the pediatric surgeon was not too keen on operating. The other mode of therapy is radiotherapy which carries variable risks of other sequel e.g., cirrhosis, hepatosarcoma and leukemia [7,8]. The non-surgical interruption can be done with hepatic artery embolization, obliteration of the vascular channels to accelerate involution by the use of steel wire, platinum coils and polyvinyl and alcohol [2,11,14,15]. These methods are messy and risky. In our case the large vascular cavities regressed from 58 x 29 x 50 mm in size to 25 x 10 x 20 mm within 3 months after transcatheter intervention. There was a suggestion that we should have used the vascular plug instead of ADO II. To block the 10 mm vessel we needed either 12 or 14 mm vascular plug which require bigger sheaths to introduce. Whereas 6 x 6 ADO II has 12 mm retention disc and went through 4F shuttle sheath. The disadvantage of ADO II is, it does not have polyester material hence PVC particles can pass through the device. Therefore we initially injected the grated gel foam, which does not pass through the device. Finally after check angiogram, before withdrawing the catheter the PVC particles, mixed with diluted contrast were injected slowly so that the particles did not reflux into the proximal arteries. There was an option to retrieve the device after the procedure. Retrospectively we were right in deploying the device in hepatic vein, because when we gave heparin infusion for the loss of femoral artery, transiently vessels opened on the abdominal ultrasound. Probably the device prevented the possible embolization of the soft thrombi from the mass.
Any neonate presenting with intractable congestive cardiac failure with persistent fetal pulmonary hypertension without lung lesion requires a high index of suspicion for diagnosis of neonatal bacterial endocarditis or IHH. The management options are varied, ranging from surgical to intervention in catheterization laboratory for IHH. Mortality is very high if left untreated, hence the need for early intervention in symptomatic infants.
4. CONCLUSION
The transcatheter device closure of hepatic vein by ADO II and injection of gel foam and PVC particles is safe and effective in IHH patient with intractable heart failure.
CONSENT
All authors declare that ‘written informed consent was obtained from the patient (or other approved parties) for publication of this case report and accompanying images.
ETHICAL APPROVAL
Not applicable
COMPETING INTERESTS
Authors have declared that no competing interests exist.
REFERENCES
- Becker JM, Heitler MS. Hepatic hemangioendotheliomas in infancy. Surg Gynecol Obstet. 1989;168:189-200.
- Fok TF, Chan MS, Metreweli C, Ng PC, Yeung CK, Li AK. Hepatic haemangioendothelioma presenting with early heart failure in a newborn: treatment with hepatic artery embolization and interferon. Acta Paediatr. 1996;85:1373-5.
- Dena M, Selby J, Thomas Stocker, Myron A, Waclawiw, Chales L, et al. Infantile hemangioendothelioma of the liver. Hepatology. 1994;20:39-45.
- Jackson C, Greene HL, O'Neill J, Kirchner S. Hepatic hemangioendothelioma: angiographic appearance and apparent prednisolone responsiveness. Am J Dis Child. 1977;131:74-7.
- Davenport M, Hansen L, Heaton ND, Howard ER. Haemangioendothelioma of the liver in infants. J Pediatr Surg. 1995;30:44-8.
- Rocchini AP, Rosenthal A, Issenberg HJ, Nadas AS. Hepatic hemangioendotheliomata: hemodynamic observations and treatment. Pediatrics. 1976;57:131-5.
- Al-Rashid RA. Cyclophosphamide and radiation therapyin the treatment of hemangioendothelioma with disseminated intravascular clotting. Cancer. 1971;27: 364-8.
- Rotman M, John M, Sowe S, Inamdar S. Radiation treatment of pediatric hepatic hemangiomatosis and coexisting cardiac failure. N Engl J Med. 1980;302:852-5.
- DeLorimier AA, Simpson EB, Baum RS, Carlsson E. Hepatic artery ligation for hepatic hemangiomatosis. N Engl J Med. 1967;277:333-7.
- Mattioli L, Lee KR, Holder TM. Hepatic artery ligation for cardiac failure due to hepatic hemangioma in the newborn. J Pediatr Surg. 1974;9:859-62.
- Enjolras O, Riche MC, Merland JJ, Escande JP. Management of alarming hemangiomas in infancy: A review of 25 cases. Pediatrics. 1990;85:491-8.
- Towes RL, Nelson JA, Hyde CT. Hepatic haemangioma successful resection in a neonate. Surgery. 1971;40:7882-5.
- Samuel M, Spitz L. Infantile hepatic hemangioendothelioma: The role of surgery. J Pediatr Surg. 1995;30:1425-9.
- Burrows PE, Rosenberg HC, Chuang HS. Diffuse hepatic hemangiomas: percutaneous transcatheter embolization with detachable silicone balloons.Radiology. 1985;156:85-8.
- Stanley P, Grinnell VS, Stanton RE, Willians KO, Shore NA. Therapeutic embolization of infantile hepatic hemangioma with polyvinyl alcohol. AJR Am J Roentgenol. 1983;141:1047-51.
Wrong title and wrong article
By : MADHAV HEGDE - On : 4/20/2018 4:46:19 PM